Earth
Expanding Roads in Africa’s Mountains Threaten Endangered Wildlife
As road networks expand into Africa’s mountainous regions, endangered and vulnerable wildlife face increasing risks of roadkill. Experts warn that without better monitoring and conservation efforts, this growing threat could decimate unique biodiversity

The remote mountain regions of the world, once pristine and largely untouched by human development, are becoming increasingly perilous for wildlife. As road networks extend into these rugged terrains, endangered and vulnerable species are facing an ominous new threat—roadkill.
Species such as the African wild dog (endangered), lions and leopards (both vulnerable), elephants (endangered), and honey badgers (near threatened) are at grave risk, according to new research presented by Professor Aliza le Roux, Assistant Dean of the Faculty of Natural and Agricultural Sciences at the University of the Free State. These animals, many classified by the International Union for Conservation of Nature (IUCN) as at risk, face an increasingly perilous existence.

In her compelling presentation at the Southern African Mountain Conference (SAMC2025), she revealed the stark reality that these once-untouched ecosystems are now being invaded by expanding roads that are leading to more wildlife-vehicle collisions, many of them fatal.
“Wildlife in these regions is incredibly vulnerable, and as roads push deeper into mountainous areas, we’re seeing a dramatic rise in roadkill incidents,” said Prof Le Roux. “Among the casualties, we’re finding not just mammals, but also critically endangered birds like the hooded vulture and the steppe eagle.”
The conference, which brought together leading researchers, policymakers, and environmental experts, focused on the dire state of mountain ecosystems, communities, and biodiversity. UNESCO, in partnership with the University of the Free State’s Afromontane Research Unit, the African Mountain Research Foundation, and the Global Mountain Safeguard Research Programme, facilitated the gathering.
For the past several months, Prof Le Roux and her colleagues—Dr Katlego Mashiane, a lecturer at the UFS Department of Geography, and Dr Clara Grilo of the BIOPOLIS project in Portugal—have scoured decades’ worth of data on roadkill, analyzing published papers from 1971 to 2024. The findings were both alarming and illuminating, revealing that the majority of roadkill data available for Africa has emerged only in the 21st century.
A Growing Threat to Wildlife
In mountainous regions, amphibians were found to be the most frequent roadkill victims, while mammals, particularly those most vulnerable to extinction, were most often killed in the low-lying regions. In some high-elevation mountains, nearly 8% of mammals killed on the roads were species classified as endangered or vulnerable. Even more alarming, the roadkill rate in these regions continues to rise as human development accelerates.
“Many smaller species—those weighing less than 1 kilogram—fall victim to vehicles simply because we don’t see them. However, larger mammals, such as elephants or antelope, are often noticed only after the crash,” Prof Le Roux explained. “When these large animals are killed, it’s not just a loss for the species; it’s a loss for us too, as these collisions can cause significant damage to vehicles and pose a danger to human drivers.”
The risk is compounded by unpredictable weather and treacherous mountain roads, where sudden changes in terrain and visibility make it difficult for both drivers and wildlife to react in time. Prof Le Roux and her team noted that the ruggedness of these areas makes it harder for animals to detect oncoming vehicles, raising the likelihood of accidents.

“These regions are already dangerous for drivers, but for wildlife, the roads are a death trap,” Prof Le Roux said. “The increased number of vehicles, combined with better-paved roads, is putting more and more wildlife at risk.”
Using sophisticated tools like Google Earth Engine’s geospatial platform, the researchers analyzed data from a variety of terrains, classifying areas by elevation. High-elevation mountains, defined as regions above 2,000 meters, moderate elevations between 1,500 and 2,000 meters, and lowland areas below 1,500 meters were all found to have significant roadkill rates, particularly among mammals and birds of conservation concern.
Data Gaps and Underreporting
The study highlighted a critical issue: limited data. Despite the alarming trends, the lack of comprehensive, systematic data across much of the African continent has left major gaps in understanding the full scope of the roadkill crisis. Data was available for only 10 countries, and much of the information consisted of ‘snapshots’ rather than long-term, continuous monitoring.
“There is so much we don’t know about the true scale of this issue across Africa, particularly in the central and western regions,” Prof Le Roux lamented. “What we do know is that these collisions are happening in areas that are home to species that exist nowhere else. We cannot afford to ignore the threat to biodiversity in these mountain ecosystems.”
As the push for infrastructure development grows, the consequences for wildlife have never been clearer. The growing mortality rate among vulnerable species, many of which are already at risk of extinction, could result in devastating losses for biodiversity.
Prof Le Roux and her colleagues urge immediate action to mitigate these risks, calling for more comprehensive data collection, better road planning, and the implementation of wildlife corridors to safeguard these precious ecosystems.
“We must recognize that as we expand our roads into these high-risk areas, we’re also taking a toll on the very creatures that make these mountains so unique,” Prof Le Roux said, emphasizing the urgent need for a balanced approach to development and conservation.
As the conference came to a close, one message echoed throughout the halls: The future of Africa’s mountainous wildlife depends on the actions we take now. The clock is ticking, and the road ahead may be the final journey for some of the continent’s most endangered creatures.
Earth
Study Reveals How Ocean Bacteria Drive Plastic Biodegradation

Plastic biodegradation is emerging as a critical solution to the global waste crisis, and new research from Massachusetts Institute of Technology offers important insights into how this process actually works in nature.
A new study by researchers at Massachusetts Institute of Technology has shed fresh light on how bacteria in marine environments collaborate to break down biodegradable plastics—offering critical insights into tackling the global plastic waste crisis.
Biodegradable plastics have long been seen as a potential solution to mounting environmental pollution. However, scientists have struggled to determine how long these materials persist in real-world conditions and how microbial communities contribute to their breakdown.
The study, published in Environmental Science and Technology, marks one of the first efforts to identify the specific roles individual bacterial species play in plastic biodegradation.
“Plastic biodegradation is highly dependent on the microbial community where the plastic ends up,” says lead author Marc Foster, a PhD researcher in the MIT-WHOI Joint Program. “It’s also dependent on the chemistry of the plastic itself.”
Unlike earlier studies that focused on single microbes, the MIT team examined how multiple bacterial species work together—a more realistic representation of how plastics degrade in nature.
The researchers studied a widely used biodegradable plastic known as an aromatic aliphatic co-polyester, commonly found in shopping bags, food packaging, and agricultural films.
Samples of this plastic were first exposed to seawater in the Mediterranean, allowing natural bacterial communities to form biofilms on their surface. Scientists then isolated and analysed these microbes in the lab.
Key findings include:
- One bacterium, Pseudomonas pachastrellae, was able to break down the plastic polymer into smaller chemical components.
- Other bacterial species were needed to consume those individual chemicals, including terephthalic acid, sebacic acid, and butanediol.
- No single bacterium could complete the entire degradation process alone.
When researchers combined five complementary bacterial species, they were able to replicate the full degradation process observed in a larger microbial community.
“This complementary function was essential,” Foster explains. “None of the bacteria alone could achieve the same level of degradation as when they worked together.”
Why Plastic Biodegradation Rates Vary
The findings suggest that the speed and efficiency of plastic biodegradation depend on several key factors:
- Microbial diversity in the environment
- Chemical composition of the plastic
- Environmental conditions such as temperature and depth
Notably, the study also found that the same bacterial community could not degrade a different type of plastic, indicating that microbial systems may be highly material-specific.
Implications for Future Plastic Recycling Solutions
The research represents an important step toward developing microbial recycling systems that could convert plastic waste into useful materials.

By understanding how bacteria interact with plastics at a molecular level, scientists could:
- Design plastics that degrade more predictably
- Engineer microbial communities for faster plastic breakdown
- Develop biological recycling technologies
Foster notes that future work will explore how to optimise bacterial combinations and improve enzyme interactions with plastic surfaces.
A Step Forward in Tackling the Plastic Waste Crisis
With more than half of global plastic waste ending up in landfills or the environment, understanding plastic biodegradation is crucial for sustainable material design.
This study highlights a fundamental shift in perspective: plastic degradation is not driven by a single organism, but by complex microbial collaboration.
As research advances, such insights could help bridge the gap between biodegradable materials and real-world environmental outcomes—bringing science closer to solving one of the planet’s most pressing pollution challenges.The study, published in Environmental Science and Technology, marks one of the first efforts to identify the specific roles individual bacterial species play in plastic biodegradation.
“Plastic biodegradation is highly dependent on the microbial community where the plastic ends up,” says lead author Marc Foster, a PhD researcher in the MIT-WHOI Joint Program. “It’s also dependent on the chemistry of the plastic itself.”
Unlike earlier studies that focused on single microbes, the MIT team examined how multiple bacterial species work together—a more realistic representation of how plastics degrade in nature.
Earth
EP Investigation: Hidden Epidemic, Tuberculosis Spreads Among Kerala’s Captive Elephants
An EP Investigation into tuberculosis in Kerala’s captive elephants reveals human transmission risks, weak screening systems, and urgent policy gaps.

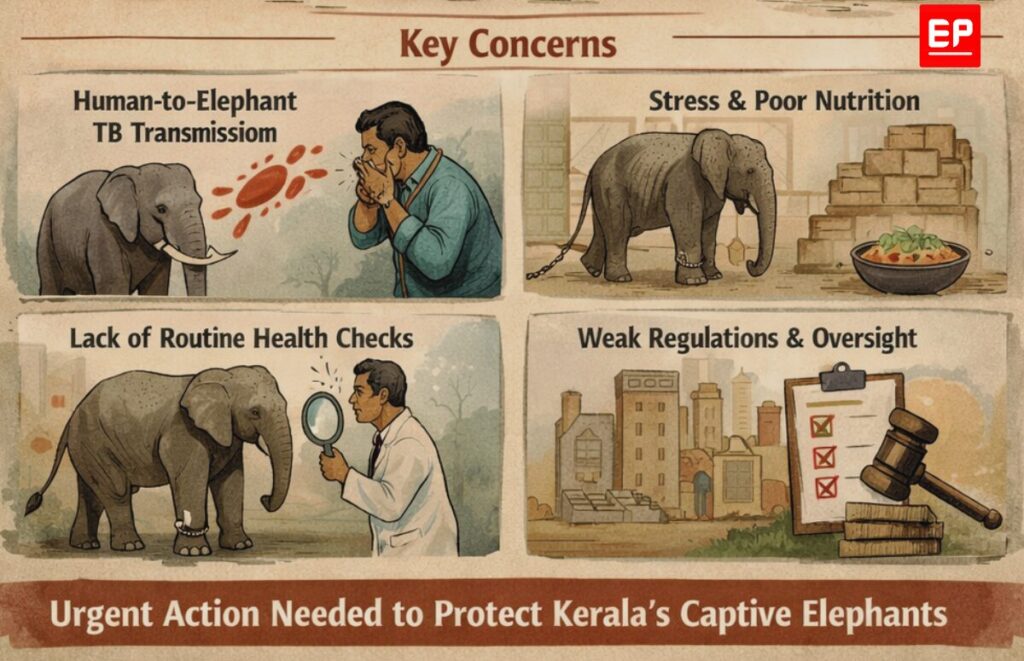
Tuberculosis in Kerala’s captive elephants has become a silent but persistent threat, driven largely by human-to-animal transmission, chronic stress, and systemic failures in veterinary public health. An EdPublica (EP) Investigation reveals how the absence of routine screening, weak governance, and prolonged neglect could turn a preventable disease into a far larger crisis in the years ahead.
By Lakshmi Narayanan | EP Investigation
Tuberculosis is quietly spreading among Kerala’s captive elephants, sustained not by wildlife exposure but by human contact, chronic stress, and systemic neglect. Long treated as a marginal veterinary issue, the disease represents a serious and largely ignored public health and animal welfare crisis—one that experts warn could intensify in the coming years if left unaddressed.
Kerala hosts one of the largest populations of captive Asian elephants in India, housed by temples, private owners, and festival organisers. According to a Forest Department survey concluded in February 2025, the state currently has 389 captive elephants, marking a steady decline from 521 in 2018 and over 700 in 2010, with the majority now owned by private individuals. This sharp reduction over the past decade reflects broader stresses within the captive elephant system, including ageing animals, declining ownership viability, and chronic health concerns.
Within this shrinking population, tuberculosis is neither new nor rare; it is endemic. Historical veterinary records and animal welfare documentation indicate that in earlier years, TB may have contributed to as many as 25 captive elephant deaths annually. Yet in recent times, detailed and transparent reporting on TB-related infections and fatalities has largely disappeared from public view, creating a misleading impression that the risk has diminished when, in reality, surveillance itself has weakened.
This absence of attention does not signal reduced risk. Tuberculosis is a slow, insidious disease that can remain latent or undiagnosed for years. Without mandatory screening or transparent surveillance, infection can circulate undetected within captive elephant populations—allowing animals to suffer prolonged illness and potentially function as silent reservoirs of infection.

The persistence of tuberculosis among captive elephants is not accidental. It is the result of a convergence of vulnerabilities: constant exposure to infected humans, immune suppression driven by captivity-related stress, and systemic failures in veterinary public health governance. Together, these factors have created ideal conditions for a preventable disease to endure—largely unseen, and largely unchallenged.
The Human–Elephant Interface: A Critical Transmission Pathway
The primary route of TB transmission among Kerala’s captive elephants is reverse zoonosis: the spread of infection from humans to animals. The causative agent, Mycobacterium tuberculosis, is a human-adapted pathogen transmitted through respiratory aerosols. In settings where elephants live and work in close proximity to people, this pathway becomes epidemiologically decisive.
Mahouts and handlers represent the most significant source of chronic exposure. Their daily routines—feeding, bathing, training, and transporting elephants—require prolonged, close physical contact. If a handler carries an active or latent TB infection, the opportunity for transmission to the animal is constant and cumulative.
In addition to handlers, the general public constitutes a secondary but important exposure source. Kerala’s festival culture routinely places elephants amid dense crowds, often for extended periods. These gatherings create intermittent but high-volume opportunities for transmission from undiagnosed or untreated individuals within the broader population. Together, these human reservoirs ensure that captive elephants are rarely insulated from the pathogen. Yet exposure alone does not fully explain disease persistence. The risk of infection is significantly magnified by conditions that undermine the elephants’ immune defenses.
“Tuberculosis in captive elephants is a severe and often underestimated disease. What is seen during post-mortem examinations is extensive, chronic organ damage that reflects prolonged suffering rather than sudden illness. These findings are consistent with long-term exposure to Mycobacterium tuberculosis and delayed detection, Dr. Arun Vishvanathan, a veterinary expert based in Kerala’s Palakkad district, tells EdPublica.
“From a medical and public health perspective, this condition is particularly concerning because it is largely driven by human-to-animal transmission. Elephants living in close, continuous contact with people—especially under stressful captive conditions—experience immune suppression, which allows the infection to progress unchecked. This is not an unavoidable disease; it is a preventable one. Without routine screening of both handlers and elephants, early diagnosis, and strict biosecurity measures, such cases will continue to occur, resulting in needless animal suffering and ongoing public health risk,” Dr. Arun Vishvanathan adds.
Stress, Captivity, and Immune Compromise
Captive environments impose profound physiological and psychological stress on elephants, a species evolved for expansive movement, complex social structures, and environmental autonomy. Confinement to restricted spaces, prolonged chaining, limited exercise, and forced participation in noisy, crowded festivals all contribute to chronic stress.
Scientific evidence across species demonstrates that sustained stress suppresses immune function. In elephants, this immunosuppression reduces resistance to opportunistic infections such as TB and increases the likelihood that latent infections will progress to active disease.
Crowding further compounds the problem. Elephants housed in close quarters or transported frequently between venues are exposed not only to more humans but also to environments conducive to airborne disease transmission. In these conditions, respiratory pathogens can spread efficiently, especially when animals are already physiologically compromised.
”Tuberculosis in Kerala’s captive elephants spreads primarily through close, repeated contact with infected humans, and is sustained by conditions that weaken the animals’ natural defenses. Unlike many wildlife diseases, this is not an infection originating in forests—it is largely a human-driven disease cycle. Mahouts and handlers are the most significant transmission source. Daily activities such as feeding, bathing, chaining, and transport require close physical proximity, often for hours at a time. If a handler has active or undiagnosed TB, the elephant is repeatedly exposed to infectious aerosols,” says Manuprasad, an elephant welfare worker from Thrissur.
Festival crowds and tourists create additional exposure. During temple festivals and public events, elephants are surrounded by dense crowds, sometimes for entire days. In these settings, even brief exposure to multiple infected individuals can result in infection.
Systemic Gaps in Veterinary Public Health
Perhaps the most critical vulnerability lies not in biology but in governance. Kerala lacks a standardized, mandatory TB screening programme for captive elephants. As a result, infected animals—many of them asymptomatic—remain undiagnosed for years. This failure in routine surveillance effectively blinds any meaningful public health response and allows elephants to function as silent reservoirs of infection.
Experts warn that tuberculosis in Kerala’s captive elephants could expand if mandatory screening and biosecurity measures are not urgently implemented.
Nutritional inadequacy is another systemic issue. Economic pressures within the temple and festival ecosystem often translate into suboptimal feeding regimes. Poor nutrition weakens immune responses, lowering the infectious dose required for TB to establish and spread.
Compounding these challenges is a widespread lack of awareness among elephant owners and handlers regarding TB transmission and prevention. Clear, enforceable biosecurity protocols—covering quarantine, treatment, and movement restrictions for TB-positive animals—are largely absent or inconsistently applied. Without such measures, even identified cases pose an ongoing risk to other elephants and to humans.

”As an animal rights and welfare activist, I have personally witnessed the post-mortem of an elephant affected by tuberculosis, and it was deeply distressing. The extent of internal damage revealed the severe and prolonged suffering this animal endured—far beyond what most people realize. Seeing such devastation in an animal of immense strength and dignity is heartbreaking,” explains Ambili Purackal, founder of DAYA, a Kerala-based NGO known for its proactive role in the state’s animal rights movement.
What makes this suffering even harder to accept is that it is largely the result of human exposure. Elephants do not face tuberculosis at these levels in the wild; they contract it through forced, prolonged contact with humans under stressful captive conditions that weaken their immunity. This is not just a veterinary concern but a moral one. These elephants are silent victims of preventable disease, and their suffering is a consequence of human neglect and systemic failure,” Ambili Purackal says.
Secondary and Less-Documented Risks
While human-to-elephant transmission remains the dominant concern, other pathways cannot be entirely dismissed. Interactions with domestic livestock or wildlife in shared environments may contribute to transmission chains, though this remains poorly documented in the Indian context. These ancillary risks further underscore the need for comprehensive epidemiological research.
A Convergence of Vulnerabilities
Taken together, the vulnerabilities facing Kerala’s captive elephants form a self-reinforcing cycle. Constant exposure to a human TB reservoir, chronic immune compromise driven by captivity-related stress and poor nutrition, and systemic failures in disease detection and control create ideal conditions for TB persistence.
Breaking this cycle will require a multi-layered public health approach—one that integrates routine screening, improved nutrition, handler health monitoring, and enforceable management protocols. Without such intervention, tuberculosis will remain a silent epidemic, exacting a slow but devastating toll on one of Kerala’s most culturally significant animal populations.
Silence, in this case, is not neutrality—it is risk.
What Needs to Change
Addressing tuberculosis among Kerala’s captive elephants requires coordinated action across animal welfare, public health, and governance. Experts and welfare workers interviewed by EdPublica point to the following urgent priorities:
1. Mandatory TB Screening
· Routine, standardised tuberculosis testing for all captive elephants
· Regular TB screening for mahouts, handlers, and caretakers
· Immediate isolation and treatment protocols for positive cases
2. Handler Health Monitoring
· Integration of mahout health checks into public TB control programmes
· Confidential diagnosis and treatment access to reduce stigma and underreporting
3. Improved Living Conditions
· Reduced chaining and confinement
· Adequate daily exercise and social interaction
· Limits on festival exposure, crowd density, and noise-related stress
4. Nutritional Standards
· Enforced minimum nutrition guidelines
· Regular veterinary audits to ensure immune-supportive diets
5. Biosecurity and Movement Controls
· Quarantine protocols for newly acquired or transferred elephants
· Restrictions on inter-district or inter-state movement of TB-positive animals
6. Transparent Reporting and Oversight
· Publicly accessible data on TB cases and outcomes
· Independent audits of temple and private elephant management practices
7. Interdepartmental Coordination
· Formal collaboration between forest, animal husbandry, and public health departments
· Recognition of TB in captive elephants as a One Health issue—linking human, animal, and environmental health
Some sources in this investigation have requested anonymity due to professional or personal safety concerns. Their identities are known to EdPublica and their statements have been independently verified.
Earth
Life may have learned to breathe oxygen hundreds of millions of years earlier than thought

Early life on Earth has found an interetsing turning point. A new study by researchers at Massachusetts Institute of Technology suggests that some of Earth’s earliest life forms may have evolved the ability to use oxygen hundreds of millions of years before it became a permanent part of the planet’s atmosphere.
Oxygen is essential to most life on Earth today, but it was not always abundant. Scientists have long believed that oxygen only became a stable component of the atmosphere around 2.3 billion years ago, during a turning point known as the Great Oxidation Event (GOE). The new findings indicate that biological use of oxygen may have begun much earlier, potentially reshaping scientists’ understanding of how life evolved on Earth.
The study, published in the journal Palaeogeography, Palaeoclimatology, Palaeoecology, traces the evolutionary origins of a key enzyme that allows organisms to use oxygen for aerobic respiration. This enzyme is present in most oxygen-breathing life forms today, from bacteria to humans.
Scientists have long believed that oxygen only became a stable component of the atmosphere around 2.3 billion years ago, during a turning point known as the Great Oxidation Event (GOE). The new findings indicate that biological use of oxygen may have begun much earlier, potentially reshaping scientists’ understanding of how life evolved on Earth
MIT geobiologists found that the enzyme likely evolved during the Mesoarchean era, between 3.2 and 2.8 billion years ago—several hundred million years before the Great Oxidation Event.
The findings may help answer a long-standing mystery in Earth’s history: why it took so long for oxygen to accumulate in the atmosphere. Scientists know that cyanobacteria, the first organisms capable of producing oxygen through photosynthesis, emerged around 2.9 billion years ago. Yet atmospheric oxygen levels remained low for hundreds of millions of years after their appearance.
While geochemical reactions with rocks were previously thought to be the main reason oxygen failed to build up early on, the MIT study suggests biology itself may also have played a role. Early organisms that evolved the oxygen-using enzyme may have consumed small amounts of oxygen as soon as it was produced, limiting how much could accumulate in the atmosphere.
“This does dramatically change the story of aerobic respiration,” said Fatima Husain, postdoctoral researcher in MIT’s Department of Earth, Atmospheric and Planetary Sciences, said in a media statement. “Our study adds to this very recently emerging story that life may have used oxygen much earlier than previously thought. It shows us how incredibly innovative life is at all periods in Earth’s history.”
The research team analysed thousands of genetic sequences of heme-copper oxygen reductases—enzymes essential for aerobic respiration—across a wide range of modern organisms. By mapping these sequences onto an evolutionary tree and anchoring them with fossil and geological evidence, the researchers were able to estimate when the enzyme first emerged.
“The puzzle pieces are fitting together and really underscore how life was able to diversify and live in this new, oxygenated world
Tracing the enzyme back through time, the team concluded that oxygen use likely appeared soon after cyanobacteria began producing oxygen. Organisms living close to these microbes may have rapidly consumed the oxygen they released, delaying its escape into the atmosphere.
“Considered all together, MIT research has filled in the gaps in our knowledge of how Earth’s oxygenation proceeded,” Husain said. “The puzzle pieces are fitting together and really underscore how life was able to diversify and live in this new, oxygenated world.”
The study adds to a growing body of evidence suggesting that life on Earth adapted to oxygen far earlier than previously believed, offering new insights into how biological innovation shaped the planet’s atmosphere and the evolution of complex life.
-

 Society3 months ago
Society3 months agoThe Ten-Rupee Doctor Who Sparked a Health Revolution in Kerala’s Tribal Highlands
-

 COP305 months ago
COP305 months agoBrazil Cuts Emissions by 17% in 2024—Biggest Drop in 16 Years, Yet Paris Target Out of Reach
-

 Society3 months ago
Society3 months agoFrom Qubits to Folk Puppetry: India’s Biggest Quantum Science Communication Conclave Wraps Up in Ahmedabad
-

 Women In Science5 months ago
Women In Science5 months agoThe Data Don’t Lie: Women Are Still Missing from Science — But Why?
-

 Earth4 months ago
Earth4 months agoData Becomes the New Oil: IEA Says AI Boom Driving Global Power Demand
-

 COP304 months ago
COP304 months agoCorporate Capture: Fossil Fuel Lobbyists at COP30 Hit Record High, Outnumbering Delegates from Climate-Vulnerable Nations
-

 Space & Physics3 months ago
Space & Physics3 months agoIndian Physicists Win 2025 ICTP Prize for Breakthroughs in Quantum Many-Body Physics
-

 Health5 months ago
Health5 months agoAir Pollution Claimed 1.7 Million Indian Lives and 9.5% of GDP, Finds The Lancet

































