The Sciences
UFS researcher tackles plastic pollution with innovative biodegradable polymers
Biodegradable polymers serve as a more environmentally friendly alternative to conventional petroleum-based plastics.

A researcher from the University of the Free State (UFS), South Africa, is making significant strides in the fight against plastic pollution through her work on biodegradable polymers—large, chain-like molecules that serve as a more environmentally friendly alternative to conventional petroleum-based plastics.
Plastic pollution has reached alarming levels globally, with an estimated 19 to 23 million tonnes of plastic waste entering aquatic ecosystems each year. Dr. Julia Puseletso Mofokeng, a Senior Lecturer and Researcher at the UFS Department of Chemistry, aims to influence both industry practices and policy decisions regarding the adoption of biodegradable polymers in disposable product packaging. “My research is aimed at managing plastic waste to combat environmental and atmospheric pollution, conserve energy, and improve water quality, including ensuring safe drinking water,” she stated.
Biodegradable polymers, derived from renewable resources like vegetable oils, starches, and animal fats, offer a sustainable alternative
According to the United Nations Environment Programme (UNEP), approximately 400 million tonnes of plastic waste are generated annually, with around 36% used for packaging—much of which ends up in landfills. Dr. Mofokeng’s research is particularly inspired by her experiences in Bophelong village in Qwaqwa, Free State, where improper waste disposal practices, including burning plastic, posed serious environmental risks.
Biodegradable polymers, derived from renewable resources like vegetable oils, starches, and animal fats, offer a sustainable alternative. “These materials can be easily disposed of after use without harming the environment,” Dr. Mofokeng explained. Her research focuses on the preparation and characterization of fully biodegradable polymer blends, which can be utilized in various applications including packaging, water purification, and electromagnetic interference shielding.
Dr. Mofokeng’s ongoing experiments involve testing three different biodegradable polymer systems under various environmental conditions to assess their degradation rates. Early signs of biodegradation, such as cracks and surface erosion, were observed after just 14 months, indicating that these polymers could completely degrade within two to three years—compared to the hundreds or thousands of years it takes for traditional plastics to break down.
The push towards biodegradable options is gaining momentum in South Africa, with many food outlets already opting for paper and bio-based materials for cutlery and packaging. “We are now left with policymakers to enforce strict laws governing production and for retail industries to adopt biopolymers in disposable packaging materials,” Dr. Mofokeng noted.
Her work aligns with the United Nations’ Sustainable Development Goals (SDGs), focusing on health and wellbeing, clean water, sustainable cities, responsible consumption, and marine conservation. With nearly two decades of experience in polymer research, Dr. Mofokeng continues to educate her community and supervise numerous students in their academic journeys.
Looking ahead, she plans to investigate the removal of heavy metals and contaminants from groundwater in Qwaqwa, aiming for practical solutions that improve water quality for local households. With the support of international collaborations and a dedicated research team, Dr. Mofokeng is determined to contribute to a more sustainable future.
Climate
Study Finds Warming Could Slightly Boost Atmosphere’s Methane-Cleaning Capacity
New research suggests climate warming may modestly enhance the atmosphere’s ability to break down methane, though competing chemical processes add uncertainty.
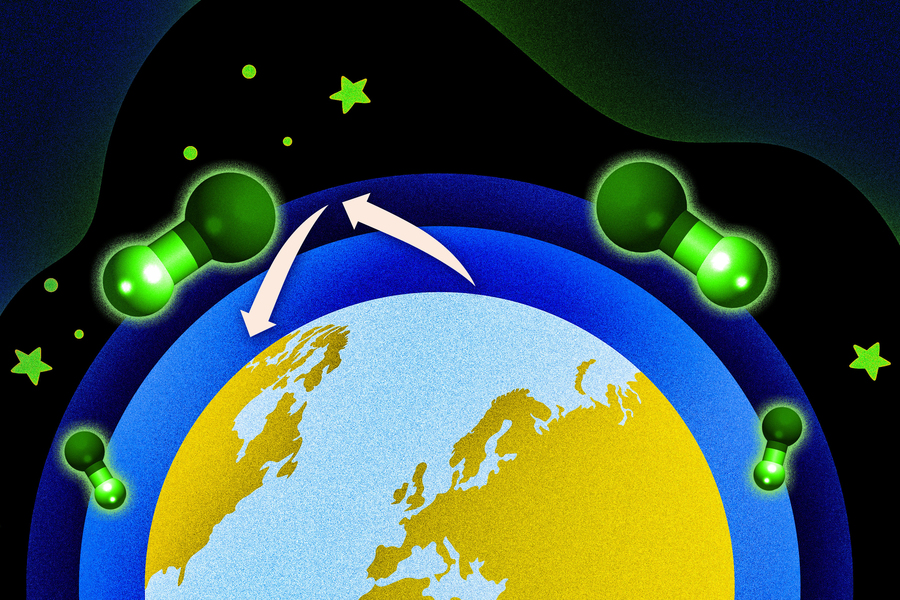
New research suggests climate warming may modestly enhance the atmosphere’s ability to break down methane, though competing chemical processes add uncertainty.
A new study by researchers at the Massachusetts Institute of Technology (MIT) finds that rising global temperatures could slightly increase the atmosphere’s ability to break down methane, one of the most potent greenhouse gases.
Methane is a major driver of global warming, second only to carbon dioxide. However, it does not persist as long in the atmosphere due to the presence of hydroxyl radicals—highly reactive molecules often described as the “atmosphere’s detergent” for their role in breaking down pollutants.
Balancing Effects of Water Vapour and Natural Emissions
The MIT team developed a new atmospheric model to understand how hydroxyl radical (OH) levels may respond to warming temperatures. Their findings reveal a complex balance of competing effects.
As global temperatures rise, atmospheric water vapour is expected to increase, boosting OH levels by about 9%. However, higher temperatures will also lead to increased emissions of natural gases from plants—known as biogenic volatile organic compounds—which can reduce OH levels by approximately 6%.
The net effect, according to the study, is a modest increase of around 3% in the atmosphere’s capacity to break down methane under a 2°C warming scenario.
Why Hydroxyl Radicals Matter
Hydroxyl radicals play a critical role in regulating atmospheric chemistry. They react with methane and other gases, breaking them down into less harmful compounds.
“About 90 percent of the methane that’s removed from the atmosphere is due to the reaction with OH,” said study author Qindan Zhu in a statement.
Beyond methane, OH also helps remove air pollutants and gases that affect public health, including ozone.
“There’s a whole range of environmental reasons why we want to understand what’s going on with this molecule,” said Arlene Fiore, a professor at MIT.
New Model Offers Deeper Insights
To conduct the study, researchers developed a model called “AquaChem,” which simulates atmospheric chemistry under different climate scenarios. The model builds on simplified “aquaplanet” systems, allowing scientists to isolate atmospheric processes without the complexity of land and ice interactions.
Using this model, the team compared current climate conditions with a scenario in which global temperatures rise by 2°C—widely considered a likely outcome without significant emissions reductions.
Uncertainty Around Natural Emissions
Despite the findings, researchers caution that there is still significant uncertainty—particularly regarding how plant emissions will respond to climate change.
Biogenic emissions, such as isoprene released by trees, appear to play a major role in influencing OH levels but remain difficult to predict accurately.
Future research will aim to refine these estimates and better understand how different climate scenarios could affect atmospheric chemistry.
Implications for Climate Projections
Even small changes in hydroxyl radical levels can have significant implications for how methane accumulates in the atmosphere.
“Understanding future trends of OH will allow us to determine future trends of methane,” Zhu said.
As methane continues to rise alongside carbon dioxide, insights into these chemical processes will be critical for improving climate models and informing mitigation strategies.
Earth
Study Reveals How Ocean Bacteria Drive Plastic Biodegradation

Plastic biodegradation is emerging as a critical solution to the global waste crisis, and new research from Massachusetts Institute of Technology offers important insights into how this process actually works in nature.
A new study by researchers at Massachusetts Institute of Technology has shed fresh light on how bacteria in marine environments collaborate to break down biodegradable plastics—offering critical insights into tackling the global plastic waste crisis.
Biodegradable plastics have long been seen as a potential solution to mounting environmental pollution. However, scientists have struggled to determine how long these materials persist in real-world conditions and how microbial communities contribute to their breakdown.
The study, published in Environmental Science and Technology, marks one of the first efforts to identify the specific roles individual bacterial species play in plastic biodegradation.
“Plastic biodegradation is highly dependent on the microbial community where the plastic ends up,” says lead author Marc Foster, a PhD researcher in the MIT-WHOI Joint Program. “It’s also dependent on the chemistry of the plastic itself.”
Unlike earlier studies that focused on single microbes, the MIT team examined how multiple bacterial species work together—a more realistic representation of how plastics degrade in nature.
The researchers studied a widely used biodegradable plastic known as an aromatic aliphatic co-polyester, commonly found in shopping bags, food packaging, and agricultural films.
Samples of this plastic were first exposed to seawater in the Mediterranean, allowing natural bacterial communities to form biofilms on their surface. Scientists then isolated and analysed these microbes in the lab.
Key findings include:
- One bacterium, Pseudomonas pachastrellae, was able to break down the plastic polymer into smaller chemical components.
- Other bacterial species were needed to consume those individual chemicals, including terephthalic acid, sebacic acid, and butanediol.
- No single bacterium could complete the entire degradation process alone.
When researchers combined five complementary bacterial species, they were able to replicate the full degradation process observed in a larger microbial community.
“This complementary function was essential,” Foster explains. “None of the bacteria alone could achieve the same level of degradation as when they worked together.”
Why Plastic Biodegradation Rates Vary
The findings suggest that the speed and efficiency of plastic biodegradation depend on several key factors:
- Microbial diversity in the environment
- Chemical composition of the plastic
- Environmental conditions such as temperature and depth
Notably, the study also found that the same bacterial community could not degrade a different type of plastic, indicating that microbial systems may be highly material-specific.
Implications for Future Plastic Recycling Solutions
The research represents an important step toward developing microbial recycling systems that could convert plastic waste into useful materials.

By understanding how bacteria interact with plastics at a molecular level, scientists could:
- Design plastics that degrade more predictably
- Engineer microbial communities for faster plastic breakdown
- Develop biological recycling technologies
Foster notes that future work will explore how to optimise bacterial combinations and improve enzyme interactions with plastic surfaces.
A Step Forward in Tackling the Plastic Waste Crisis
With more than half of global plastic waste ending up in landfills or the environment, understanding plastic biodegradation is crucial for sustainable material design.
This study highlights a fundamental shift in perspective: plastic degradation is not driven by a single organism, but by complex microbial collaboration.
As research advances, such insights could help bridge the gap between biodegradable materials and real-world environmental outcomes—bringing science closer to solving one of the planet’s most pressing pollution challenges.The study, published in Environmental Science and Technology, marks one of the first efforts to identify the specific roles individual bacterial species play in plastic biodegradation.
“Plastic biodegradation is highly dependent on the microbial community where the plastic ends up,” says lead author Marc Foster, a PhD researcher in the MIT-WHOI Joint Program. “It’s also dependent on the chemistry of the plastic itself.”
Unlike earlier studies that focused on single microbes, the MIT team examined how multiple bacterial species work together—a more realistic representation of how plastics degrade in nature.
Earth
Life may have learned to breathe oxygen hundreds of millions of years earlier than thought

Early life on Earth has found an interetsing turning point. A new study by researchers at Massachusetts Institute of Technology suggests that some of Earth’s earliest life forms may have evolved the ability to use oxygen hundreds of millions of years before it became a permanent part of the planet’s atmosphere.
Oxygen is essential to most life on Earth today, but it was not always abundant. Scientists have long believed that oxygen only became a stable component of the atmosphere around 2.3 billion years ago, during a turning point known as the Great Oxidation Event (GOE). The new findings indicate that biological use of oxygen may have begun much earlier, potentially reshaping scientists’ understanding of how life evolved on Earth.
The study, published in the journal Palaeogeography, Palaeoclimatology, Palaeoecology, traces the evolutionary origins of a key enzyme that allows organisms to use oxygen for aerobic respiration. This enzyme is present in most oxygen-breathing life forms today, from bacteria to humans.
Scientists have long believed that oxygen only became a stable component of the atmosphere around 2.3 billion years ago, during a turning point known as the Great Oxidation Event (GOE). The new findings indicate that biological use of oxygen may have begun much earlier, potentially reshaping scientists’ understanding of how life evolved on Earth
MIT geobiologists found that the enzyme likely evolved during the Mesoarchean era, between 3.2 and 2.8 billion years ago—several hundred million years before the Great Oxidation Event.
The findings may help answer a long-standing mystery in Earth’s history: why it took so long for oxygen to accumulate in the atmosphere. Scientists know that cyanobacteria, the first organisms capable of producing oxygen through photosynthesis, emerged around 2.9 billion years ago. Yet atmospheric oxygen levels remained low for hundreds of millions of years after their appearance.
While geochemical reactions with rocks were previously thought to be the main reason oxygen failed to build up early on, the MIT study suggests biology itself may also have played a role. Early organisms that evolved the oxygen-using enzyme may have consumed small amounts of oxygen as soon as it was produced, limiting how much could accumulate in the atmosphere.
“This does dramatically change the story of aerobic respiration,” said Fatima Husain, postdoctoral researcher in MIT’s Department of Earth, Atmospheric and Planetary Sciences, said in a media statement. “Our study adds to this very recently emerging story that life may have used oxygen much earlier than previously thought. It shows us how incredibly innovative life is at all periods in Earth’s history.”
The research team analysed thousands of genetic sequences of heme-copper oxygen reductases—enzymes essential for aerobic respiration—across a wide range of modern organisms. By mapping these sequences onto an evolutionary tree and anchoring them with fossil and geological evidence, the researchers were able to estimate when the enzyme first emerged.
“The puzzle pieces are fitting together and really underscore how life was able to diversify and live in this new, oxygenated world
Tracing the enzyme back through time, the team concluded that oxygen use likely appeared soon after cyanobacteria began producing oxygen. Organisms living close to these microbes may have rapidly consumed the oxygen they released, delaying its escape into the atmosphere.
“Considered all together, MIT research has filled in the gaps in our knowledge of how Earth’s oxygenation proceeded,” Husain said. “The puzzle pieces are fitting together and really underscore how life was able to diversify and live in this new, oxygenated world.”
The study adds to a growing body of evidence suggesting that life on Earth adapted to oxygen far earlier than previously believed, offering new insights into how biological innovation shaped the planet’s atmosphere and the evolution of complex life.
-

 Society3 months ago
Society3 months agoThe Ten-Rupee Doctor Who Sparked a Health Revolution in Kerala’s Tribal Highlands
-

 COP305 months ago
COP305 months agoBrazil Cuts Emissions by 17% in 2024—Biggest Drop in 16 Years, Yet Paris Target Out of Reach
-

 Society3 months ago
Society3 months agoFrom Qubits to Folk Puppetry: India’s Biggest Quantum Science Communication Conclave Wraps Up in Ahmedabad
-

 Women In Science5 months ago
Women In Science5 months agoThe Data Don’t Lie: Women Are Still Missing from Science — But Why?
-

 Earth4 months ago
Earth4 months agoData Becomes the New Oil: IEA Says AI Boom Driving Global Power Demand
-

 COP304 months ago
COP304 months agoCorporate Capture: Fossil Fuel Lobbyists at COP30 Hit Record High, Outnumbering Delegates from Climate-Vulnerable Nations
-

 Space & Physics3 months ago
Space & Physics3 months agoIndian Physicists Win 2025 ICTP Prize for Breakthroughs in Quantum Many-Body Physics
-

 Health5 months ago
Health5 months agoAir Pollution Claimed 1.7 Million Indian Lives and 9.5% of GDP, Finds The Lancet








































