The Sciences
IIT Ropar unveils eco-friendly mechanical machine for knee rehabilitation
The introduction of this innovative mechanical CPM machine marks a significant step toward democratizing healthcare and improving rehabilitation outcomes globally.

In a major development for knee rehabilitation, researchers at Indian Institute of Technology (IIT) Ropar have introduced a revolutionary, low-cost solution to make Continuous Passive Motion (CPM) therapy more accessible to patients. The team’s newly patented innovation, the Completely Mechanical Passive Motion Machine for Knee Rehabilitation, is set to transform post-surgical recovery, especially in resource-limited areas.
Unlike traditional motorized CPM devices, which are expensive and reliant on electricity, the new machine operates entirely through mechanical means. Utilizing a piston and pulley system that stores air as the user pulls a handle, the device enables smooth, controlled knee motion to aid in rehabilitation. This design eliminates the need for electricity, batteries, or motors, making the machine lightweight, portable, and environmentally friendly.
The mechanical CPM machine addresses a key barrier to knee therapy: the high cost and power dependence of conventional electric machines. It offers a viable alternative for patients, particularly in rural and off-grid areas, where access to electricity is often unreliable. Its portability also enables patients to continue their therapy at home, reducing the need for frequent hospital visits or prolonged stays.
Knee rehabilitation is crucial for patients recovering from surgeries, as CPM therapy helps improve joint mobility, reduce stiffness, and speed up recovery. With this new device, IIT Ropar’s researchers are offering a cost-effective, sustainable option that could improve the lives of countless patients, especially in India, where advanced medical technology can be scarce in rural regions.
Lead researcher Dr. Abhishek Tiwari, along with his team members Suraj Bhan Mundotiya and Dr. Samir C. Roy, expressed optimism about the machine’s potential. “This device has the power to revolutionize knee rehabilitation, particularly in areas where access to sophisticated medical equipment is limited. It’s designed to be an affordable and eco-friendly solution that not only aids in recovery but also minimizes environmental impact,” said Dr. Tiwari.
The introduction of this innovative mechanical CPM machine marks a significant step toward democratizing healthcare and improving rehabilitation outcomes globally.
The Sciences
Scientists Crack the Genetic Code Behind Seedless Grapes
Indian researchers reveal how genetic mutations create the popular seedless varieties consumers love, opening doors to better grape breeding

Scientists have finally unlocked the mystery behind one of our favourite fruit features – seedless grapes. A study by Indian researchers has decoded the genetic mechanisms that create seedless grapes, potentially revolutionizing how we breed these popular fruits.
The Science Behind Seedless Success
The research, led by Dr. Ravindra Patil at the Agharkar Research Institute (ARI) in Pune, India, examined why some grapes develop without seeds while others don’t. Published in BMC Plant Biology, the study focused on a seedless mutant derived from the high-yielding grape variety ARI-516.
Using advanced genomic tools, the team discovered that pollen sterility is the key culprit behind seedlessness. The seedless grapes showed:
- Abnormal pollen structure with very low viability
- Complete inability of pollen grains to germinate
- Smaller female reproductive structures compared to seeded varieties
- Disrupted fertilization processes leading to seedless berry formation
Genetic Detective Work
The researchers employed cutting-edge techniques to crack the code:
Transcriptomic Analysis: RNA sequencing revealed that genes crucial for pollen development, cell division, and hormone signaling were significantly “switched off” in seedless varieties.
Whole-Genome Sequencing: This identified multiple insertion-deletion mutations (InDels) in genes responsible for pollen development, essentially breaking the normal seed formation process.
The evidence points to parthenocarpy – a natural process where fruits develop without fertilization due to reproductive defects.
Why This Matters
This discovery isn’t just academic curiosity. Seedless grapes represent a massive global market, with consumers strongly preferring them for:
- Fresh consumption
- Raisins and dried products
- Juices and processed foods
The research provides molecular markers that grape breeders can use to:
- Develop new seedless varieties faster
- Improve fruit quality and yield
- Create grapes better adapted to different climates
Impact on Agriculture
Dr. Patil’s team has conducted one of the most comprehensive studies on grape seedlessness using modern genomic tools. This breakthrough could significantly accelerate breeding programs worldwide, benefiting both grape growers and consumers who enjoy these convenient, seed-free fruits.
The study represents a perfect example of how understanding nature’s genetic mechanisms can lead to practical agricultural improvements, potentially transforming grape cultivation and meeting growing global demand for high-quality seedless varieties.
The research was conducted in collaboration with Savitribai Phule Pune University and represents a significant advancement in horticultural genomics.
Climate
Study Finds Warming Could Slightly Boost Atmosphere’s Methane-Cleaning Capacity
New research suggests climate warming may modestly enhance the atmosphere’s ability to break down methane, though competing chemical processes add uncertainty.
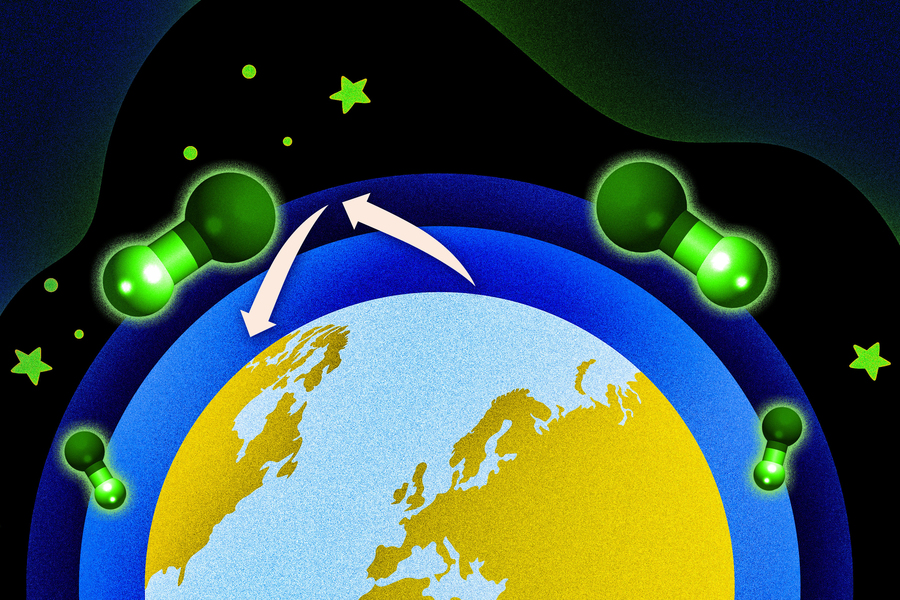
New research suggests climate warming may modestly enhance the atmosphere’s ability to break down methane, though competing chemical processes add uncertainty.
A new study by researchers at the Massachusetts Institute of Technology (MIT) finds that rising global temperatures could slightly increase the atmosphere’s ability to break down methane, one of the most potent greenhouse gases.
Methane is a major driver of global warming, second only to carbon dioxide. However, it does not persist as long in the atmosphere due to the presence of hydroxyl radicals—highly reactive molecules often described as the “atmosphere’s detergent” for their role in breaking down pollutants.
Balancing Effects of Water Vapour and Natural Emissions
The MIT team developed a new atmospheric model to understand how hydroxyl radical (OH) levels may respond to warming temperatures. Their findings reveal a complex balance of competing effects.
As global temperatures rise, atmospheric water vapour is expected to increase, boosting OH levels by about 9%. However, higher temperatures will also lead to increased emissions of natural gases from plants—known as biogenic volatile organic compounds—which can reduce OH levels by approximately 6%.
The net effect, according to the study, is a modest increase of around 3% in the atmosphere’s capacity to break down methane under a 2°C warming scenario.
Why Hydroxyl Radicals Matter
Hydroxyl radicals play a critical role in regulating atmospheric chemistry. They react with methane and other gases, breaking them down into less harmful compounds.
“About 90 percent of the methane that’s removed from the atmosphere is due to the reaction with OH,” said study author Qindan Zhu in a statement.
Beyond methane, OH also helps remove air pollutants and gases that affect public health, including ozone.
“There’s a whole range of environmental reasons why we want to understand what’s going on with this molecule,” said Arlene Fiore, a professor at MIT.
New Model Offers Deeper Insights
To conduct the study, researchers developed a model called “AquaChem,” which simulates atmospheric chemistry under different climate scenarios. The model builds on simplified “aquaplanet” systems, allowing scientists to isolate atmospheric processes without the complexity of land and ice interactions.
Using this model, the team compared current climate conditions with a scenario in which global temperatures rise by 2°C—widely considered a likely outcome without significant emissions reductions.
Uncertainty Around Natural Emissions
Despite the findings, researchers caution that there is still significant uncertainty—particularly regarding how plant emissions will respond to climate change.
Biogenic emissions, such as isoprene released by trees, appear to play a major role in influencing OH levels but remain difficult to predict accurately.
Future research will aim to refine these estimates and better understand how different climate scenarios could affect atmospheric chemistry.
Implications for Climate Projections
Even small changes in hydroxyl radical levels can have significant implications for how methane accumulates in the atmosphere.
“Understanding future trends of OH will allow us to determine future trends of methane,” Zhu said.
As methane continues to rise alongside carbon dioxide, insights into these chemical processes will be critical for improving climate models and informing mitigation strategies.
Earth
Study Reveals How Ocean Bacteria Drive Plastic Biodegradation

Plastic biodegradation is emerging as a critical solution to the global waste crisis, and new research from Massachusetts Institute of Technology offers important insights into how this process actually works in nature.
A new study by researchers at Massachusetts Institute of Technology has shed fresh light on how bacteria in marine environments collaborate to break down biodegradable plastics—offering critical insights into tackling the global plastic waste crisis.
Biodegradable plastics have long been seen as a potential solution to mounting environmental pollution. However, scientists have struggled to determine how long these materials persist in real-world conditions and how microbial communities contribute to their breakdown.
The study, published in Environmental Science and Technology, marks one of the first efforts to identify the specific roles individual bacterial species play in plastic biodegradation.
“Plastic biodegradation is highly dependent on the microbial community where the plastic ends up,” says lead author Marc Foster, a PhD researcher in the MIT-WHOI Joint Program. “It’s also dependent on the chemistry of the plastic itself.”
Unlike earlier studies that focused on single microbes, the MIT team examined how multiple bacterial species work together—a more realistic representation of how plastics degrade in nature.
The researchers studied a widely used biodegradable plastic known as an aromatic aliphatic co-polyester, commonly found in shopping bags, food packaging, and agricultural films.
Samples of this plastic were first exposed to seawater in the Mediterranean, allowing natural bacterial communities to form biofilms on their surface. Scientists then isolated and analysed these microbes in the lab.
Key findings include:
- One bacterium, Pseudomonas pachastrellae, was able to break down the plastic polymer into smaller chemical components.
- Other bacterial species were needed to consume those individual chemicals, including terephthalic acid, sebacic acid, and butanediol.
- No single bacterium could complete the entire degradation process alone.
When researchers combined five complementary bacterial species, they were able to replicate the full degradation process observed in a larger microbial community.
“This complementary function was essential,” Foster explains. “None of the bacteria alone could achieve the same level of degradation as when they worked together.”
Why Plastic Biodegradation Rates Vary
The findings suggest that the speed and efficiency of plastic biodegradation depend on several key factors:
- Microbial diversity in the environment
- Chemical composition of the plastic
- Environmental conditions such as temperature and depth
Notably, the study also found that the same bacterial community could not degrade a different type of plastic, indicating that microbial systems may be highly material-specific.
Implications for Future Plastic Recycling Solutions
The research represents an important step toward developing microbial recycling systems that could convert plastic waste into useful materials.

By understanding how bacteria interact with plastics at a molecular level, scientists could:
- Design plastics that degrade more predictably
- Engineer microbial communities for faster plastic breakdown
- Develop biological recycling technologies
Foster notes that future work will explore how to optimise bacterial combinations and improve enzyme interactions with plastic surfaces.
A Step Forward in Tackling the Plastic Waste Crisis
With more than half of global plastic waste ending up in landfills or the environment, understanding plastic biodegradation is crucial for sustainable material design.
This study highlights a fundamental shift in perspective: plastic degradation is not driven by a single organism, but by complex microbial collaboration.
As research advances, such insights could help bridge the gap between biodegradable materials and real-world environmental outcomes—bringing science closer to solving one of the planet’s most pressing pollution challenges.The study, published in Environmental Science and Technology, marks one of the first efforts to identify the specific roles individual bacterial species play in plastic biodegradation.
“Plastic biodegradation is highly dependent on the microbial community where the plastic ends up,” says lead author Marc Foster, a PhD researcher in the MIT-WHOI Joint Program. “It’s also dependent on the chemistry of the plastic itself.”
Unlike earlier studies that focused on single microbes, the MIT team examined how multiple bacterial species work together—a more realistic representation of how plastics degrade in nature.
-

 Society4 months ago
Society4 months agoThe Ten-Rupee Doctor Who Sparked a Health Revolution in Kerala’s Tribal Highlands
-

 Society4 months ago
Society4 months agoFrom Qubits to Folk Puppetry: India’s Biggest Quantum Science Communication Conclave Wraps Up in Ahmedabad
-

 COP306 months ago
COP306 months agoBrazil Cuts Emissions by 17% in 2024—Biggest Drop in 16 Years, Yet Paris Target Out of Reach
-

 Earth6 months ago
Earth6 months agoData Becomes the New Oil: IEA Says AI Boom Driving Global Power Demand
-

 COP305 months ago
COP305 months agoCorporate Capture: Fossil Fuel Lobbyists at COP30 Hit Record High, Outnumbering Delegates from Climate-Vulnerable Nations
-

 Space & Physics4 months ago
Space & Physics4 months agoIndian Physicists Win 2025 ICTP Prize for Breakthroughs in Quantum Many-Body Physics
-

 Health6 months ago
Health6 months agoAir Pollution Claimed 1.7 Million Indian Lives and 9.5% of GDP, Finds The Lancet
-

 Sustainable Energy5 months ago
Sustainable Energy5 months agoThe $76/MWh Breakthrough: Battery-Backed Solar Becomes the Cheapest Firm Power





























